Polarity of Dopamine: To determine if the molecule is polar or none polar, the electronegativity of each bond must be calculated. Since dopamine is a larger molecule; calculating the electronegativity is not easily done.
Steps for determining polarity:
Intermolecular Bonds within Dopamine: These are the forces that govern all physical properties of molecules: boiling point, melting point, viscosity, surface tension etc. There exists a force called London dispersion forces which exists in all molecules. These forces are simply the attraction between positive and negative charges. In dopamine there exist three different kinds of intermolecular bonds.
The Role of Electrons in Dopamine: The electron configuration in molecules determine the structural geometry of that molecule. In dopamine there are many dipole moments therefore the structure is not linear, since their charges cannot balance each other. There are also five lone pairs, 2 each on oxygen and one on nitrogen. Lone pairs tend to be very negative which can make dopamine very attractive or even repulsive at its ends. Many proteins in the blood stream contain a slightly negative charge which causes dopamine to flow nicely through the blood stream without being restricted, since the protein and dopamine molecules will slightly repel each other. This type of electron structure is very efficient as it can reach it transmitters easily. Dopamine though is not polar enough to cross the brain blood barrier. This is so because the benzene ring of carbons that hold the dopamine together form a symmetrical pattern. Electrons in the benzene ring will cancel each other making it nonpolar at that point which renders dopamine useless to passing the barrier, which is great because dopamine needs to react in the brain centers. Note that dopamine is still polar because it is not linear in shape and contains many dipole moments.
Reactions Involving Dopamine: Unlike most molecules in chemistry, dopamine does not react directly react with the substances that trigger it. Instead it reacts with neurotransmitters within the brain. Say after you eat some food with sugar, the sugar will activate the sensors in the basal ganglia which will release dopamine in the bloodstream. Dopamine molecules then travels to the nucleus accumbens and striatum where they interlock with specific transmitters like a lock and key device.
Steps for determining polarity:
- Determining the electronegativity (the ability of an atom to attract electrons to itself in a chemical bond). The electronegativity of hydrogen is a mere 2.1 while carbon, nitrogen, and oxygen, have 2.5, 3.0, and 3.5 respectively.
- Find the electronegative difference. The concept states that if there is a difference of (0-0.3) it is covalent, (0.4-1.9) it is polar covalent, and 2.0+ is ionic. There are six main elemental bonds in Dopamine, H-O, O-C, C-C, C-H, N-C, and N-H.
- O-H: 3.5-2.1=1.4. (Polar covalent)
- O-C: 3.5-2.5=1.0. (Polar covalent)
- C-C: 2.5-2.5=0. (Nonpolar)
- C-H: 2.5-2.1=0.4. (Polar covalent)
- N-C: 3.0-2.5=0.5. (Polar covalent)
- N-H: 3.0-2.1=0.9. (Polar covalent)
- Find the net dipole moment of each bond which occurs any time there is a separation of positive and negative charges.
Intermolecular Bonds within Dopamine: These are the forces that govern all physical properties of molecules: boiling point, melting point, viscosity, surface tension etc. There exists a force called London dispersion forces which exists in all molecules. These forces are simply the attraction between positive and negative charges. In dopamine there exist three different kinds of intermolecular bonds.
- London Dispersion Forces: As mentioned before is the coulombic attraction between atoms. Example of this in dopamine is prominent within all atoms.
- Dipole-dipole Forces: Occurs when two polar molecules attract each other. One element will be slightly positive and the other will be slightly negative hence the attraction. Example of this in dopamine is C-H.
- Hydrogen Bonding: This is the same as dipole-dipole forces, except on steroids. This occurs when hydrogen is bonded to Oxygen, Nitrogen, and Flourine. These forces are stronger than regular dipole-dipole forces because hydrogen has a tiny positive charge compared to the largely negative charge of N,O,and F. In dopamine hydrogen is bonded to oxygen and nitrogen, making it very strong intermolecularly.
The Role of Electrons in Dopamine: The electron configuration in molecules determine the structural geometry of that molecule. In dopamine there are many dipole moments therefore the structure is not linear, since their charges cannot balance each other. There are also five lone pairs, 2 each on oxygen and one on nitrogen. Lone pairs tend to be very negative which can make dopamine very attractive or even repulsive at its ends. Many proteins in the blood stream contain a slightly negative charge which causes dopamine to flow nicely through the blood stream without being restricted, since the protein and dopamine molecules will slightly repel each other. This type of electron structure is very efficient as it can reach it transmitters easily. Dopamine though is not polar enough to cross the brain blood barrier. This is so because the benzene ring of carbons that hold the dopamine together form a symmetrical pattern. Electrons in the benzene ring will cancel each other making it nonpolar at that point which renders dopamine useless to passing the barrier, which is great because dopamine needs to react in the brain centers. Note that dopamine is still polar because it is not linear in shape and contains many dipole moments.
Reactions Involving Dopamine: Unlike most molecules in chemistry, dopamine does not react directly react with the substances that trigger it. Instead it reacts with neurotransmitters within the brain. Say after you eat some food with sugar, the sugar will activate the sensors in the basal ganglia which will release dopamine in the bloodstream. Dopamine molecules then travels to the nucleus accumbens and striatum where they interlock with specific transmitters like a lock and key device.
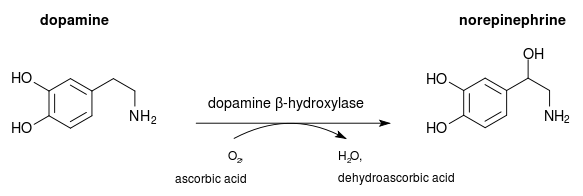
Dopamine to Norephedrine:
Dopamine being such a large molecule does not react with much other molecules to form distinctively different compounds like the combustion of methane for example. Instead the end of the molecule, its organic functional group reacts with other elements or small molecules to form a different compound with similar properties.
Dopamine being such a large molecule does not react with much other molecules to form distinctively different compounds like the combustion of methane for example. Instead the end of the molecule, its organic functional group reacts with other elements or small molecules to form a different compound with similar properties.

Dopamine to L-DOPA:

Dopamine and HCl:
When dopamine is synthesized within a laboratory it needs to be mixed with a fixed proportion of HCl to be stable at room temperature and readily available for injection. This of course is a chemical reaction.
C8H11NO2(aq) + HCl (aq)----> C8H12ClNO2 (aq)
When dopamine is synthesized within a laboratory it needs to be mixed with a fixed proportion of HCl to be stable at room temperature and readily available for injection. This of course is a chemical reaction.
C8H11NO2(aq) + HCl (aq)----> C8H12ClNO2 (aq)
Dopamine as a Limiting Reactant:
As mentioned before, dopamine does not react directly react with any substance. Therefore it being a limiting reactant in a reaction is not very easy to find. But in life, from the beginning to the end it can act as a limiting reactant to the body itself in terms of movement. Lack of dopamine within the basal ganglia can cause a disease called "Parkinson's disease". Whenever the neurons of the brain die or become impaired they produce less dopamine which can inhibit fluent and smooth movements that was before an easy task to achieve. Also abusive cocaine habits which causes excess dopamine to be produced can cause the neurons to die faster as they are using more energy than normal.
As mentioned before, dopamine does not react directly react with any substance. Therefore it being a limiting reactant in a reaction is not very easy to find. But in life, from the beginning to the end it can act as a limiting reactant to the body itself in terms of movement. Lack of dopamine within the basal ganglia can cause a disease called "Parkinson's disease". Whenever the neurons of the brain die or become impaired they produce less dopamine which can inhibit fluent and smooth movements that was before an easy task to achieve. Also abusive cocaine habits which causes excess dopamine to be produced can cause the neurons to die faster as they are using more energy than normal.
Structure in Correlation to Dopamine's properties:
- Because the structure is not liner it gives the the molecule a certain degree of polarity.
- There are 5 lone pairs of electrons at the very ends of the molecule which give dopamine a slightly negative charge at the ends. This helps the molecule to navigate quite easily through the bloodstream since proteins are slightly negative also. So they will repel each other and not cause any friction.
- The benzene ring located in the center of the molecule is symmetrical in shape therefore non polar. Even though dopamine is polar overall, this benzene ring makes it slightly less polar than other molecules and hence it cannot pass the blood brain barrier.
References:
Cover picture: http://images.fineartamerica.com/images-medium-large/2-dopamine-3d-molecular-model-pasieka.jpg
Determining the Polarity of Dopamine: Chemistry A Molecular Approach by Nivaldo J. Tro.
http://courses.washington.edu/conj/bess/intro/intro3.html
Intermolecular Bonds within Dopamine: http://dopaminerulezxo.weebly.com/bond-analysis.html
Chemistry A Molecular Approach by Nivaldo J. Tro.
The Role of Electrons in Dopamine: http://chemwiki.ucdavis.edu/Theoretical_Chemistry/Chemical_Bonding/Lewis_Theory_of_Bonding/Geometry_of_Molecules
http://www.quora.com/Why-cant-dopamine-cross-the-blood-brain-barrier-while-L-dopa-or-other-substituted-phenethylamines-can
Picture of dopamine and Transmitters:
http://www.humanillnesses.com/images/hdc_0000_0001_0_img0085.jpg
https://31.media.tumblr.com/a42b2bce00a6916310b8d9b6851d05a7/tumblr_inline_mxw932ZlN01qhg0wt.gif
Reactions of Dopamine:
http://upload.wikimedia.org/wikipedia/commons/thumb/8/8b/Dopamine_beta-hydroxylase_reaction.svg/583px-Dopamine_beta-hydroxylase_reaction.svg.png
http://3.bp.blogspot.com/_B4FBju9poGc/S3Nx-8ROf9I/AAAAAAAAANA/6xAvBL4t-vQ/s400/tyrosine+ADHD+L-DOPA+to+dopamine+DOPA+decarboxylase.PNG
http://www.chemspider.com/Chemical-Structure.58820.html
Dopamine as a Limiting reactant:
http://nihseniorhealth.gov/parkinsonsdisease/whatcausesparkinsonsdisease/01.html
Cover picture: http://images.fineartamerica.com/images-medium-large/2-dopamine-3d-molecular-model-pasieka.jpg
Determining the Polarity of Dopamine: Chemistry A Molecular Approach by Nivaldo J. Tro.
http://courses.washington.edu/conj/bess/intro/intro3.html
Intermolecular Bonds within Dopamine: http://dopaminerulezxo.weebly.com/bond-analysis.html
Chemistry A Molecular Approach by Nivaldo J. Tro.
The Role of Electrons in Dopamine: http://chemwiki.ucdavis.edu/Theoretical_Chemistry/Chemical_Bonding/Lewis_Theory_of_Bonding/Geometry_of_Molecules
http://www.quora.com/Why-cant-dopamine-cross-the-blood-brain-barrier-while-L-dopa-or-other-substituted-phenethylamines-can
Picture of dopamine and Transmitters:
http://www.humanillnesses.com/images/hdc_0000_0001_0_img0085.jpg
https://31.media.tumblr.com/a42b2bce00a6916310b8d9b6851d05a7/tumblr_inline_mxw932ZlN01qhg0wt.gif
Reactions of Dopamine:
http://upload.wikimedia.org/wikipedia/commons/thumb/8/8b/Dopamine_beta-hydroxylase_reaction.svg/583px-Dopamine_beta-hydroxylase_reaction.svg.png
http://3.bp.blogspot.com/_B4FBju9poGc/S3Nx-8ROf9I/AAAAAAAAANA/6xAvBL4t-vQ/s400/tyrosine+ADHD+L-DOPA+to+dopamine+DOPA+decarboxylase.PNG
http://www.chemspider.com/Chemical-Structure.58820.html
Dopamine as a Limiting reactant:
http://nihseniorhealth.gov/parkinsonsdisease/whatcausesparkinsonsdisease/01.html
